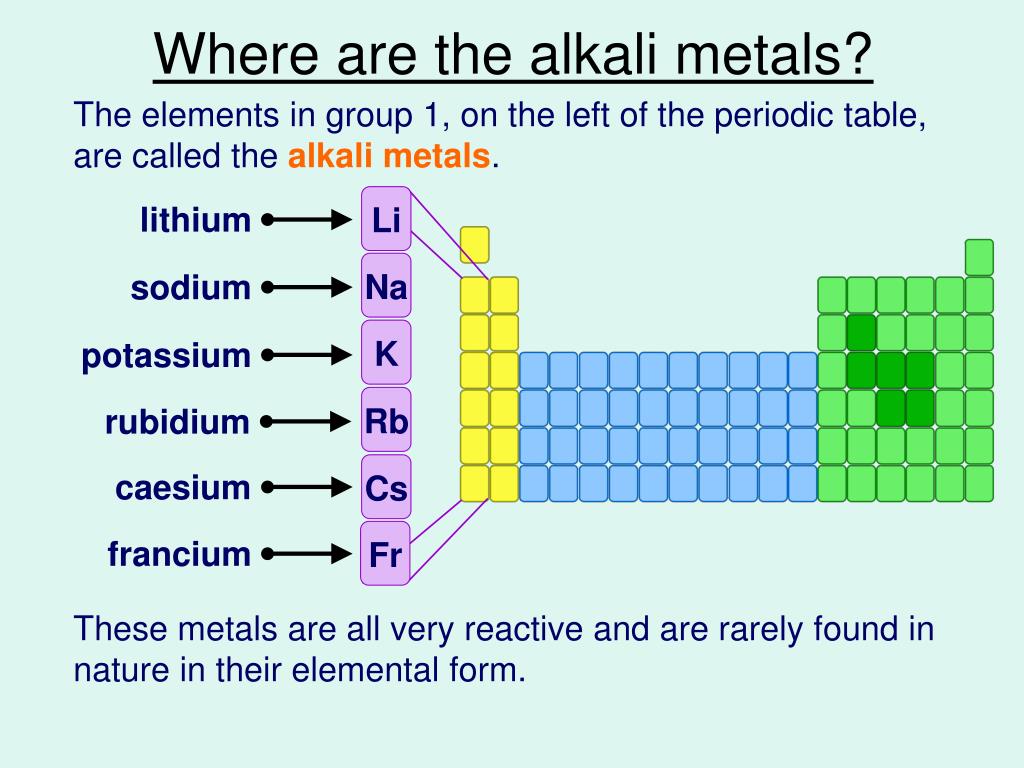
Chemical Reactivity decrease as you go down the groupįor Non-Metals, the farther right-up in the table you go, the higher the electronegativity.Chemical Reactivity increases as you go left to right of the periodic table.And you go from left to right on the periodic table elements have more electrons in their valence shells they have to get rid off which requires an element to have high energies which results into lower Chemical Reactivity. This happens because as you go down a group, it is easier for electrons to be taken or given away, resulting in high Chemical Reactivity. Chemical Reactivity increases as you go down the group.Chemical Reactivity decrease as you go left to right of the periodic table.Īnother factor is the Electronegativity of the element, which determine how badly an element wants to take other electrons from other elements. One factor is Ionization Energy, Ionization Energy is how easily and element can remove its electrons. Pearson Education South Asia Pte Ltd 2005.A couple factors will determine how likely and element will react to another element. Longman Pocket Study Guide 'O' Level Science-Chemistry by Lim Eng Wah Chapter 8 pg 190.Pearson Education South Asia Pte Ltd 2005. Science in Focus, Chemistry for GCE 'O' Level by J G R Briggs Chapter 11 pg 172.Only a metal higher in the reactivity series will displace another. Therefore the elemental metal will 'displace' the ionic metal over time, thus the two swap places. When a metal in elemental form is placed in a solution of a metal salt it may be, overall, more energetically feasible for this "elemental metal" to exist as an ion and the "ionic metal" to exist as the element. However it is defined by the nature of the metals in single displacement reactions. The reactivity series determines qualitatively characteristics such as the reactions with water, air and acids as demonstrated above. does not readily give up electrons in reactions to form positive ions.does not react vigorously and quickly with chemicals.readily gives up electrons in reactions to form positive ionsĪ metal 'low down' in the reactivity series:. 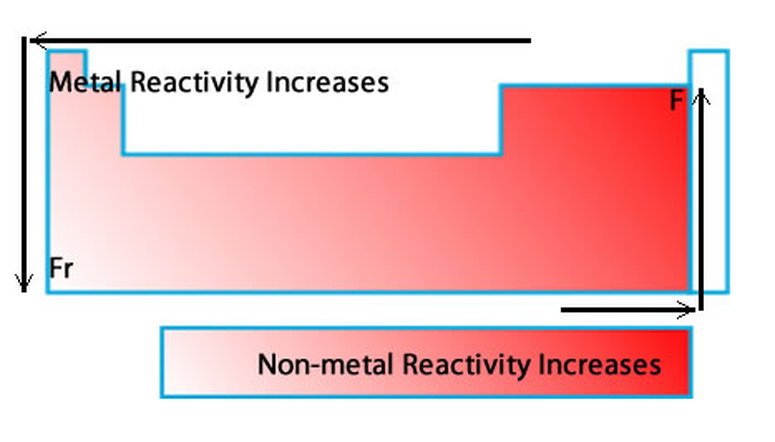
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
